| Main Index New Window Search Profile Send Private Check Private Who's Online Elements Table FAQ No Encryption Song Logout | ||||
| 32 Online, 4493 Registered | You have 0 new messages | |||
 
All posts in this forum will be visible only after moderator approval. |
| Serious Chemistry | Previous thread Forum index Threaded Next thread | ||
| All 24 posts | ||
| hest
(Hive Bee) 04-19-01 11:07 No 185131 |
New Amph. more potent than LSD | Reply | ||||||
In J.Med.Chem. 2001,44, 1003-1010, there is an article abouth some very potent sub. amphetamines The TM is this one (pic 1) [ 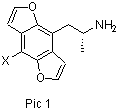 they both make the R form and the S form For thr most potent form, the R form, the strenght is: When x is H the A is a bit stronger than DOB When x is Br the A is app. 10-20 times stronger than DOB When x is CF3 the A is app. 5-10 times stronger than DOB The S form of the TM has app. the ½ strenght of the R form (that's still a lot:) The synt in the article start with l alanine, and introduce the opt.act. center from the beginning. Nice but hard to do at home. But if we start with this one, it become much easyer [ .gif) All the steps is standardt chem. The following steps should also bee posible [ .gif) Mabye the bromination should bee the last thing to doo. The oxidation with DDQ is also posible with NBS or Pd/C in wacum (dehydrogenation). It is this oxidation that makes the TM so potent, the non oxidated molecule has also been made (J.Med.Chem.1996,39,2953-2961) Some bioassayes on human would bee nice, but i'l guess we have to do the hard work :-) |
||||||||
| Rhodium (Chief Bee) 04-19-01 20:43 No 185224  |
Re: New Amph. more potent than LSD | Reply | ||||||
Great find! The bromination cannot be done as the last step, or the furan rings will be brominated. Are all the steps in the synth of the benzo-bisdihydrofuran in the article? It seems a lot better than their original synth using bromochloroethane and BuLi... http://rhodium.lycaeum.org/ |
||||||||
| hest
(Hive Bee) 04-19-01 21:32 No 185229 |
Re: New Amph. more potent than LSD | Reply | ||||||
Yes. all the steps is in the article, if 'the beehive' wants it i'l make a nice writup after the weg. |
||||||||
| savestheday
(Hive Bee) 04-20-01 00:37 No 185262 .gif) |
Re: New Amph. more potent than LSD | Reply | ||||||
Very interesting... anymore information would be greatly appreciated. Shulgin doesn't mention anything close to this? |
||||||||
| Lilienthal (Moderator) 04-20-01 03:08 No 185293 |
Re: New Amph. more potent than LSD | Reply | ||||||
See also Post No 122784 for a previous discussion on this topic  . . |
||||||||
| yellium
(Hive Bee) 04-20-01 06:13 No 185323 |
Re: New Amph. more potent than LSD | Reply | ||||||
Another question is whether it is an interesting compound. F'r instance, the 2C-G compounds are not that potent, but they are very long lasting and don't seem to be very much `fun', compared to other compounds. |
||||||||
| moo
(Stranger) 04-20-01 19:56 No 185431 |
Re: New Amph. more potent than LSD | Reply | ||||||
But then again, receptor binding and resistance to metabolism are two different stories. The reports on DOTFM make me scared. understanding is everything |
||||||||
| Rhodium (Chief Bee) 04-21-01 01:07 No 185476  |
Re: New Amph. more potent than LSD | Reply | ||||||
Everybody refers to scare stories with DOTFM, but I have never heard the actual stories. Tell me? Dosage, duration, effects? http://rhodium.lycaeum.org/ |
||||||||
| moo
(Stranger) 04-23-01 20:17 No 186117 |
Re: New Amph. more potent than LSD | Reply | ||||||
It should have said "rumours". I'm referring to what drone told, but cannot be sure. I bet certain researchers we all know could provide more information on the subject. understanding is everything |
||||||||
| hest
(Hive Bee) 04-25-01 06:08 No 186514 |
Re: New Amph. more potent than LSD | Reply | ||||||
The fist step is posible, but i gues i'll have to tune it a bit :-) [ .gif) The reaction was made in boiling methanol with NaOH as the base, reactiontime 2 h yeald 20% (yak) I will try some other solvents and a longer reaktionstime |
||||||||
| Rhodium (Chief Bee) 04-25-01 22:31 No 186707  |
Re: New Amph. more potent than LSD | Reply | ||||||
Perhaps you should try using 2-bromoethanol instead of chloroethanol? Boiling ethylene oxide in 48% HBr should produce 2-bromoethanol. KOH is probably better than NaOH too. Have you looked up any journal references on the synthesis of your product? It would probably help alot. http://rhodium.lycaeum.org/ |
||||||||
| Osmium (Stoni's sexual toy) 04-25-01 23:53 No 186728 |
Re: New Amph. more potent than LSD | Reply | ||||||
In the older ref about this class of compounds they used 1,2-bromo-chloro-ethane, or 1,2-bromo-iodo-ethane, and had higher yields. |
||||||||
| Rhodium (Chief Bee) 04-26-01 12:52 No 186901  |
Re: New Amph. more potent than LSD | Reply | ||||||
Yes, but considering the price of bromochloroethane, and the special handling of the required BuLi in the former procedure, this alternative sounds better for most of us. http://rhodium.lycaeum.org/ |
||||||||
| hest
(Hive Bee) 04-29-01 05:51 No 187650 |
Re: New Amph. more potent than LSD | Reply | ||||||
Disolve 6,16g(110mmol) KOH in 60mL wather, let the wather cool down ( 30°C), then add 5g(45mmol) hydroquinon and 8,85g(110mmol) 2-chloro-ethanol. Reflux the solution for 12 houers, cool it down (with ice) filter off the raw product. Disolve the raw product in 100mL hot EtOAC, ekstract with 3*30mL 2M NaOH, dry the EtOAC with MgSO4, and remove the solvent in vaco. yeald 3,5g (40%) pure on TLC (Rf 0,44 with EtOAC as the solvent) The same eksp. was done with 10g hydroquinon, and the yeald was again 40% I think this is the way, the yeald is not high, but the starting materials is almost free. I'll will now carye on withe the next step. |
||||||||
| yellium
(Hive Bee) 04-29-01 08:49 No 187690 |
Re: New Amph. more potent than LSD | Reply | ||||||
Do you dare to ingest the final product? |
||||||||
| smiley_boy
(Hive Bee) 04-29-01 13:47 No 187760 |
Re: New Amph. more potent than LSD | Reply | ||||||
The thing that sticks out in my mind about this is that the bromo-substituted analog is more potent in this series than its trifluoromethyl counterpart. I didn't see any discussion on this point in the paper, but it certanly warrants some sort of comment. Curious. |
||||||||
| hest
(Hive Bee) 04-29-01 21:30 No 187845 |
Re: New Amph. more potent than LSD | Reply | ||||||
yellium- This might bee the new kitchen table 'lsd' synthesis (simple (compared to lysergacid) to manufacture, stabel product and strong enough to distribute at blotters). I think i'll firs try the phenetylamin, and a methoxy group instead of the Br would proberly make the product lighter and easyer for the metabolism to degrade. |
||||||||
| yellium
(Hive Bee) 04-29-01 21:47 No 187849 |
Re: New Amph. more potent than LSD | Reply | ||||||
And without any substituent you also have a pretty active compound. |
||||||||
| weasil
(Hive Bee) 04-30-01 07:24 No 187933 |
Re: New Amph. more potent than LSD | Reply | ||||||
NO, I strongly demand you to only produce the brominated product. To do otherwise would be undermining my authority. |
||||||||
| Rhodium (Chief Bee) 05-11-01 10:26 No 193388  |
Re: New Amph. more potent than LSD | Reply | ||||||
A VERY useful reference for the synthesis of the molecule below can be found in the journal Huaxue Shiji 15(4), 246-247 (1993). Unfortunately, I have no idea at all where to find that journal, or in what language it is written. Damn CAS Online, I don't want to be reminded of excellent reactions which are located in unreachable journals. .gif)
http://rhodium.lycaeum.org/ |
||||||||
| dildoque
(Newbee) 05-11-01 19:22 No 193458 |
Re: New Amph. more potent than LSD | Reply | ||||||
I found something: Huaxue-shiji (Chemical Reactions) : shuangyuekan / Huaxue-shiji Bianjibu bianji. - Beijing : Huaxue Huaxue Shiji Keji Qingbao Zhongxinzhan, 1979- ISSN: 0258-3283  |
||||||||
| hest
(Hive Bee) 05-24-01 03:02 No 196477 |
Re: New Amph. more potent than LSD | Reply | ||||||
Subject: Re: New Amph. more potent than LSD Well, the chlorination of the alcohol was not that easye. Boiling it with SOCl2 for 12 h. did nothing. Guess you hawe to use pyridine as well ( they doo that in the article ) But i can't stand the smell, so this was the next idea [ .gif)
It works werrye well. Here is the cookbok A mixture off 11g(0,1mol) hydroquinone, 16g(0,4mol) NaOH in 80mL wather was mixet with 2,2g(0,001mol) Benzyltriethylammoniumchlorid and 0,52g NaHSO3 in 80mL 1,2dichloroethane. The mixture was refluxet for 12 houers, made acid with HCl(aq). The org layer was sep. from the wather and the 1,2-DCE was removed in wac. The solid mass was then rextalised from ethanol (250mL) Yeald app. 80%. Pure on TLC Rf 0,68 1:1 ether:petr.ether The org. phase is a 'mudy' one, but thats ok. The ammonium compound is a phase transpher catalyst. almost anny org ammonium salt works, (tetrabutylammoniumbromid and so on) It should also bee posible to doo the reaction without the PTC. the NaHSO3 makes the hydroquinone more stabel, but is not a must. Some refs Synthesis 1988 p.950-952 Cannadian j.chem. 1955 p.1442-1447 (withouth PTC) |
||||||||
| Rhodium (Chief Bee) 05-24-01 03:41 No 196490  |
Re: New Amph. more potent than LSD | Reply | ||||||
Is this procedure from an article, or from SWIM's own experience? I don't get why the hydroquinone only reacts with the dichloroethane, and not with any 2-chloroethoxyphenyl-something forming di/polymers? Is it due to the large excess of DCE? Why the NaHSO3, an antioxidant for the hydroquinone? http://rhodium.lycaeum.org/ |
||||||||
| hest
(Hive Bee) 05-24-01 03:54 No 196496 |
Re: New Amph. more potent than LSD | Reply | ||||||
From swims own experience, but the ide is from the article in synthesis. (they doo it on o-bromo-phenol) The NaHSO3 is an antioxidant. And yes, when the sodiumsalt off hydroquinon is transferet to the DCE with help from the ammonium thing, the large excess off DCE make polimer formation unlikley. The ethanol, afther the xtalisation is a bit brown, so I gues that some polimers is formed. I will go on with the bromination. And i'v seen that the aromatisation is posible with NBS (yes). You brominate the hydrofuran and then eliminate HBr in one step due to the energi won by the aromatisation. |
||||||||